27. November 2019
Fragment-Based Drug Discovery

Carna Biosciences Newsletter Vol. 5
High Throughput Screening (HTS) has been heavily relied upon to search for such lead compounds. Recently however, Fragment-Based Drug Discovery (FBDD), which is a relatively new technology to identify potential fragments under 300 Da and optimize them into drugs, has been a topic of much focus in the field. In 1996, Fesik introduced FBDD as an alternative approach to HTS for the first time in the literature (1).
One desirable quality of a compound library is high completeness, in other words large chemical space occupancy (2) . Chemical space is a concept in cheminformatics referring to the three-dimensional space constructed with predicted virtual molecules made up of the chemical elements represented, their number, and combination. The smaller the compounds that make up the library, the more comprehensive the chemical library can be. This means that the probability of finding effective compounds is higher in a fragment library consisting of small compounds. Hann reported that small and simple compounds have a lower probability of non-specific interaction with proteins and a higher probability of specific interaction with a target protein, compared to large and complex compounds (3). This implies a high chance of success using fragment screening. Small lead fragments generally fulfill the criteria of Lipinski’ s “Rule of Five” , thus lending themselves to the ability to expand lead fragments synthetically with a high degree of freedom while maintaining desirable drug-like physical and chemical properties. However, the physiological inhibitory activity of small fragments are too weak to detect using conventional assay methods like ELISA.
How to identify suitable Fragments
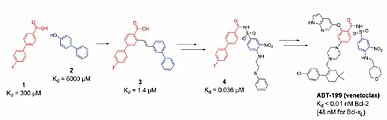
To identify binding fragments, it is often necessary to combine a number of biophysical measurement systems such as NMR, X-ray crystallography, thermal shift assay, and SPR, thus obtaining diversified information. The throughput of these techniques is inherently low. Venetoclax, a Bcl-xL inhibitor launched as a treatment for chronic lymphocytic leukemia, is a successful example of a drug discovery program that started from fragments. Venetoclax was created by connecting two fragments (Fragment 1 and Fragment 2 in Fig. 1) discovered by FBDD with a linker, then optimizing them (4). The Kd values of Fragment 1 and Fragment 2 were several hundred to several thousand μM, but the Kd value of Venetoclax was improved to 48 nM.
Weak Affinity Chromatography (WAC™)
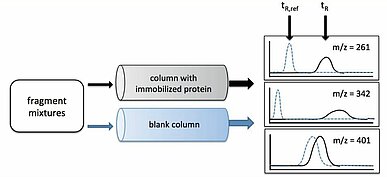
Carna is excited to introduce the benefits of unique and proprietary Weak Affinity Chromatography (WAC™) fragment screening services to its customers. This methodology is based on the combination of passing libraries of fragments over a protein covalently immobilized on a standard high-performance liquid chromatography (HPLC) column, and mass spectrometry. The KD value is directly calculated from the difference of retention time between the column with immobilized protein and the control, empty column (Fig. 2). Since the fragments eluted from the column are directly detected, the throughput is high and it is possible to measure 3,000-4,000 fragments per week. The results of WAC are reported to highly correlate with those from NMR and SPR, at 88% and 83%, respectively (5). WAC makes it possible to identify fragments with a wide range of affinity, from 0.1μM to 10mM, and decreases the necessary sample protein amount to 1/5-1/10 of other methods, while at the same time having a lower cost than SPR or X-ray crystallography. Services are offered using our 1,300-fragment library for full screening, or you can have the flexibility to utilize your own fragment library.
Please don’ t hesitate to contact us for Weak Affinity Chromatography (WAC™ ) Fragment Screening or further information or if you have questions.
References:
- Science. 1996; 274(5292):1531-4. Fesik SW.
- akugaku Zasshi, 2010; 130(3) 315-323. Tanaka D.
- J Chem Inf Comput Sci. 2001; 41(3):856-64. Hann MM.
- Essays Biochem. 2017; 61(5):475-484. Price AJ.
- Anal Chem. 2013; 85(14):6756-66. Meiby E.

Further news
-
22-02-24
Cancer treatments using inhibitors of CDK, a cell cycle regulator
Read moreCarna Newsletter Vol.16
-
22-02-23
ALK drug resistant mutations: Challenges for the treatment of lung cancer
Read moreCarna Newsletter Vol.15
-
13-10-22
2023 Year-End Campaign
Read moreWe’ve got good news for you: a special discount for human Kinases from Carna Biosciences.
