05. July 2022
The potential of PIKfyve inhibitor as a treatment of amyotrophic lateral sclerosis (ALS)

Carna Newsletter Vol.13
Amyotrophic lateral sclerosis (ALS)
ALS is a fatal neurodegenerative disease characterized by the selective and progressive degeneration of motor neurons in the brain, brainstem and spinal cord. Motor neuron deterioration leads to muscle weakness and results in death of the patient due to respiratory failure, typically within 3 to 5 years after diagnosis. The lifetime risk of developing ALS is estimated to be ~1 in 400¹⁾. Currently, two palliative treatments, riluzole and edaravone, have been approved by FDA, however their efficacy is limited and there are no effective approved treatments ²⁾.
An intronic hexanucleotide tandem repeat expansion in C9ORF72
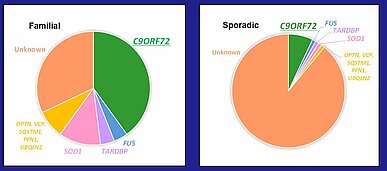
Approximately 10% of ALS is classified as familial, whereas the remaining 90% of cases are considered sporadic. An intronic hexanucleotide (GGGGCC) tandem repeat expansion was identified in the C9ORF72 gene, which is the most common genetic mutation in ALS found in ~40% of familial ALS and ~7% of sporadic ALS cases in European ancestry ³⁾ (Fig.1). While healthy individuals usually harbor up to 24 units of the hexanucleotide repeat in C9ORF72, ALS patients have more than 24, and sometimes up to hundreds or even thousands repeat units ⁴⁾.
Loss- and gain-of-function C9ORF72 mechanisms
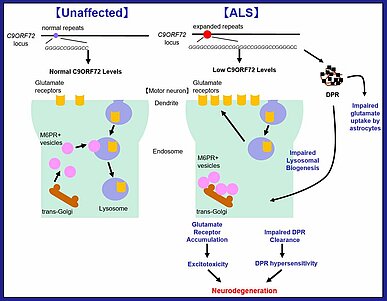
The hexanucleotide repeat expansion in C9OFR72 contributes to neurodegeneration by both loss-of-function (decreased C9ORF72 protein levels) and gain-of-function (dipeptide repeat proteins (DPRs) production derived from the hexanucleotide repeat expansion) mechanisms⁵⁾ (Fig.2).
Reduced C9ORF72 levels lead to fewer lysosomes in motor neurons, and this may be due in part to altered trafficking of endosomal vesicles and Mannose-6-Phosphate Receptor (M6PR)⁺ vesicles. Defects in lysosomal biogenesis leads to the accumulation of glutamate receptors on the cell surface and increased susceptibility to glutamate (a neurotransmitter)-induced excitotoxicity. Additionally, the clearance of DPRs which have been shown to induce toxicity is impaired⁵⁾.
As a gain-of-function mechanism, DPRs are generated from the transcripts of the C9ORF72 gene containing GGGGCC repeats by repeat-associated non-AUG translation. DPRs induce aberrant splicing and dysfunction of EAAT2 glutamate transporter in astrocytes and reduce their rate of glutamate uptake⁵⁾⁶⁾. Aberrant splicing and loss of EAAT2 protein were observed in the motor cortex and spinal cord of ALS patients, and this leads to increased extracellular glutamate and excitotoxic neuronal degeneration⁷⁾. In addition, DPRs reduced the survival of induced motor neurons (iMN) which are driven from iPS cells and importantly, C9ORF72+/-, C9ORF72-/- and C9ORF72 patient iMNs survival were reduced significantly faster than controls in response to DPRs⁵⁾.
These data suggest that loss- and gain-of-function C9ORF72 mechanisms by the hexanucleotide repeat expansion can cooperate to cause neurodegeneration in ALS, and that a drug developed to replace C9ORF72 function may be required.
Inhibition of PYKfyve with small molecules rescues the neurodegenerative mechanisms in cellular and animal ALS-models
In 2018, Shi, Y., et al. performed a phenotypic screen to identify small molecules that resulted in survival of C9ORF72 ALS patient-derived iMN. Compounds targeting diverse cellular processes including vesicle trafficking were screened. YM201636, which is a selective inhibitor of PIKfyve, a phosphatidylinositol-5-kinase, was identified as an inhibitor that significantly increased C9ORF72 patient iMN survival. Additionally, another PIKfyve inhibitor, apilimod showed similar data⁵⁾.
PIKfyve inhibition prevents the conversion of phosphatidylinositol 3-phosphate (PI3P), a membrane lipid, into phosphatidylinositol (3,5)-bisphosphate (PI(3,5)P2) and increases PI3P levels. PI3P drives early endosomal maturation and also drives lysosomal fusion with endosomes and autophagosomes. Thus, specific PIKfyve inhibition may compensate for reduced C9ORF72 activity by increasing PI3P levels to facilitate removal of glutamate receptors or DPRs⁵⁾. Providing further support for the PIKfyve importance, loss-of-function mutations in a phosphoinositide 5-phosphatase, FIG4, which has the opposite function to PIKfyve (by converting PI(3,5)P2 to PI3P), has been observed in 2% of ALS-patients and may act as a risk factor to ALS⁸⁾.
It has been reported that apilimod rescues both loss-and gain-of-function C9ORF72 disease mechanisms in murine models of C9ORF72 ALS⁹⁾.
Development of PIKfyve inhibitors for ALS treatment
The confidence within the ALS research community that the inhibition of PIKfyve may result in a disease modifying outcome for C9ORF72 patients is demonstrated by the progress of clinical trials initiated and planned by at least 2 companies. AI Therapeutics is currently recruiting for a Phase2a study for the drug LAM-002A (apilimod dimesylate), where they are hoping to measure improvements in ALS symptoms as well as indicative biomarkers. Verge Genomics is expected to enter the clinic with their PIKfyve inhibitor in 2022.
With strong evidence from patient-derived cell lines, animal models, and ALS patients that PIKfyve is an attractive target for the development of new ALS therapeutics, more researchers are exploring small molecules that could target its inhibition.
Carna’s tools to help your PIKfyve research
Carna can assist your PIKfyve research by providing protein kinases and biochemical & cellular assay services for this and other targets. Our kinases, including PIKfyve and related products, are manufactured in-house at Carna and delivered to you after stringent quality control. Carna offers a PIKfyve assay kit and a biochemical activity-based assay service using our own PIKfyve active kinase, as well as a cell-based binding assay service based on target engagement of PIKfyve using the NanoBRET™ technology.
If you are interested in Carna Biosciences’ products & services, please contact us at info@carnabio.com or our European distributor Kinase Logistics Europe at info@kinaselogistics.com.
More information on available products and services can be found using the links below.
- Lipid Kinase Proteins
- QuickScout Screening Assist™ ADP-Glo™ Assay Kits (Lipid Kinases)
- Activity-based Biochemical Profiling Assay Services (Lipid Kinases and others )
- NanoBRET™ TE Intracellular Kinase Assay Services
References:
- Brain. 2020; 143(6): 1651-1673. Guo W.
- Med Res Rev. 2019; 39(2): 733-748. Jaiswal MK.
- Nat Neurosci. 2014; 17(1): 17-23. Renton AE.
- Curr Opin Genet Dev. 2017; 44: 117-124. Van Mossevelde S.
- Nat Med. 2018; 24(3): 313-325. Shi Y.
- Science. 2014; 345(6201): 1139-45. Kwon I.
- Neuron. 1998; 20(3): 589-602. Lin CL.
- Am J Hum Genet. 2009; 84(1): 85-8. Chow CY.
- bioRxiv. 2019, doi: https://doi.org/10.1101/685800, Staats KA.

Further news
-
13-10-22
2023 Year-End Campaign
Read moreWe’ve got good news for you: a special discount for human Kinases from Carna Biosciences.
-
06-09-22
DGKα and DGKζ are key targets for cancer immunotherapy
Read moreCarna Newsletter Vol.14
-
11-06-22
New Cellular CDK Assay Panel Launched and Special Offers from Carna
Read moreWe are pleased to launch a new CDK (cyclin-dependent kinases) Cell-based Assay Panel for kinase researchers who are focusing on the CDK family.
…
