Newsletter
Residence time and compound selectivity

Carna Biosciences Newsletter Vol. 2
Human kinases are well-studied targets and have extensive involvement in a variety of diseases. More than 50 registered small molecule drugs target this enzyme class, and more than 250 kinase inhibitors are being actively evaluated in clinical trials. Most kinase inhibitors are classified as Type I, which act by binding to the ATP binding site of a kinase. While these molecules tend to exhibit strong efficacy by inhibiting many targets, this quality can also lead to adversarial effects including high toxicity caused by inhibiting off-target kinases. This negative aspect of Type I inhibitors can be acceptable in the treatment of life-threatening diseases like cancer, however next generation drug discovery and development and the treatment of more chronic conditions will require potent kinase inhibition combined with minimum side effects. In the current kinase inhibitor exploration field, target indications have been expanding well beyond cancers, where strong efficacy needs to be accompanied with high selectivity and low toxicity. Currently, the residence time (RT) of an inhibitor binding to a kinase has attracted attention as an important indicator of its efficacy and selectivity.
Evaluation of molecular interactions based on surface plasmon resonance (SPR) detection
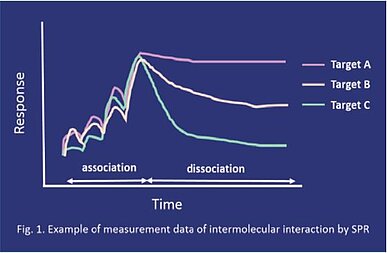
One major method to measure RT is to evaluate its molecular interactions based on the surface plasmon resonance (SPR) detection principle. This simple interaction experiment involves immobilizing one molecule of a binding pair ("ligand") on a sensor chip surface and then injecting its partner solution ("analyte") at a series of concentrations across the chip surface to detect its binding status, followed by injecting a buffer to measure its dissociation. Figure 1 shows an inhibitor having a longer RT toward target A compared to target C. When RT of an inhibitor is longer for the target kinase, and shorter for a non-target kinase, the inhibitor can be expected to be highly potent and selective toward the target kinase. Understanding characteristics of inhibitors by studying RT can help differentiate one inhibitor from another with potentially less desirable therapeutic effect. Among the 4 PI3K isoforms (α, β, γ, δ), γ and δ are expressed in the synovial membrane of patients with rheumatoid arthritis and are involved in this immunity disorder. The γ/δ selective inhibitor, duvelisib is reported to have more than 20x longer RT for PI3Kγ and PI3Kδ compared to the previous generation selective inhibitor, idelalisib, the latter having been approved for the treatment of hematologic tumors. It is also reported that the RT ratios of PI3Kγ and PI3Kδ to α, γ/α and δ/α of duvelisib are approximately 10 times higher than that of idelalisib(1). Based on these data, duvelisib was expected to be more effective and selective than idelalisib, and after observed good efficacy against arthritis model mice(2), it advanced to clinical trials for rheumatoid arthritis indications.
Carna Biosciences offers products and services to support Residence Time evaluation and recognizes RT’ s evolving role as an essential element in drug development. The services and products related to SPR and Residence Time are listed below:
- NanoBRET™ TE Intracellular Kinase Cell-Based Assay Services
- Biotinylated Kinases(available for SPR data acquisition)
Please don’ t hesitate to contact us for further information or if you have questions.
*Reference: 1) J Mol Biol. 2017; 429(4):574-586. Willemsen-Seegers N.

Further news
-
Newsletter
Cancer treatments using inhibitors of CDK, a cell cycle regulator
Read moreCarna Newsletter Vol.16
-
Newsletter
ALK drug resistant mutations: Challenges for the treatment of lung cancer
Read moreCarna Newsletter Vol.15
-
Technical Note
DGKα and DGKζ are key targets for cancer immunotherapy
Read moreCarna Newsletter Vol.14
